
A cloudy day at the Berkeley Marina.
Hidden away beneath the numerous schooners and yawls, my lab mate and I trod steadily towards a small patch of sulfurous sediment at the Berkeley Marina. With a shovel in one hand and a bright orange bucket in tow, we burrow away at the bay-soaked sediment, gradually heaping shovelfuls of pungent mud into our pail. After an hour of ambitiously digging, we wrestle our container back to the lab. What inspired a pair of scientists to leave the lab on a cloudy Autumn day to go and scoop dirt? We are microbial prospectors, and we study how life survives in nature’s nooks and crannies. For us, deep sediment harbors interesting life that survives without oxygen and that uses a mixture of distinct chemicals to breathe.
One of these chemicals, known as iodate, lingers in trace quantities throughout the world’s oceans and is a variety of iodine that is often unusable by most living creatures. Over the past decade, studies from several labs identified distinctive bacterial life that converts iodate to a more bioavailable form of iodine called iodide. Astoundingly, these bacteria do not just convert iodate to iodide as part of their life cycle; they, in fact, require iodate to grow, and produce iodide as waste! Not much is known about these microorganisms, which is why we are interested in studying how they breathe iodate and change it to a form that most mammals, seaweeds, and marine life need for survival.
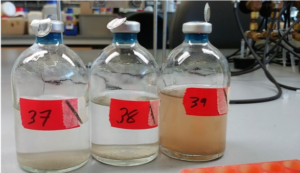
An example of the miniature microbial ecosystems, full of nutrients change color from clear (two left bottles) to reddish-brown (right) due to the removal of iodate from the liquid.
In the lab, we use multiple approaches to study these bizarre microbes. Our first approach is to dutifully cultivate the bacteria in bottles without oxygen and pack these bottles with nutrients and salts from their natural habitat. We then place spoonfuls of sediment into these clear glass bottles and flush out all the oxygen with nitrogen gas and carbon dioxide. After removing all the oxygen, a small rubber plug on the bottles keeps all the oxygen out while the bottles of microbes flourish in a warm room for several weeks.
With time, these miniature ecosystems begin to get extremely cloudy, produce gas bubbles, and sometimes even change colors. These dramatic behaviors are a testament to the extensive diversity of metabolisms and bacterial species that populate these miniature environments. To investigate the significance of these strange observations, we take samples from each of the bottles for our experiments. We then examine these samples for simple attributes, such as color or cloudiness, and for more complex attributes such as the changing concentration of iodate and iodide in the sample.
If we want to study the fascinating biology of these organisms more closely, we can also separate the bacteria from our small micro environments and examine them individually. We begin by pouring our culture into a gel-like substance with nutrients so that they remain still as they grow. After several days, these microbes start forming round spheres, called colonies, which we will pick and grow in liquid with salts and nutrients. Doing this allows us to then separate the DNA from the unknown bacteria and have their genomes sequenced. Not much is known about these bacteria, which makes the process of sequencing their genomes exhilarating. Doing so unlocks the core set of never before seen instructions that dictate how these microbes survive in these distinct environments. Most importantly, these genes will ultimately tell us how these bacteria thrive and grow in the presence of iodate.

An example of the gel-like substance changing colors from clear to a dark brown color as colonies (black dots in gel) grow inside.
Upon finding these genes and understanding the mechanisms associated with the conversion of iodate to iodide, we may then be able to identify where in the broader environment these microorganisms exist. Although we can isolate these bacteria from sediment, it could be that these microbes are more common in other locations that scientists have not studied thoroughly. The overarching purpose of this project strives to learn where and how these genes exist in the broader world, and whether other microbes use these genes to survive as well.
More directly, one may ask: How exactly does knowing all this impact us? By understanding the phenomenon of iodate conversion to iodide we begin to learn about how this element influences human health more broadly. To date, no single definitive mechanism explains how this element enters iodine rich organisms (such as seaweeds) and then into our food chain. The identification of a new group of bacteria, capable of transforming iodate to iodide may inevitably be the missing piece to that puzzle.
Understanding the movement of this element in our ecosystems will also enable us to protect humans. One prominent example would be the mitigation of radioactive isotopes that contaminate watersheds and oceans after a nuclear accident. Equally as important, learning about this elemental cycle provides a new perspective on the connectedness of our world; serving as a reminder that even the smallest life forms can contribute to phenomena important to mammalian health.
Victor Reyes-Umaña is a third-year doctoral candidate from the Coates Lab in the Department of Plant and Microbial Biology. After graduating in 2010 with his BS in Biology at the University of Edinburgh, he returned home to San Francisco to start his career in cell culture and fermentation at a local biotech company. His passion for environmental microbiology inspired his current doctoral research into microbial metabolisms and biogeochemical cycles in the environment.